|
4/30/2023 0 Comments Prophase 1 of meiosis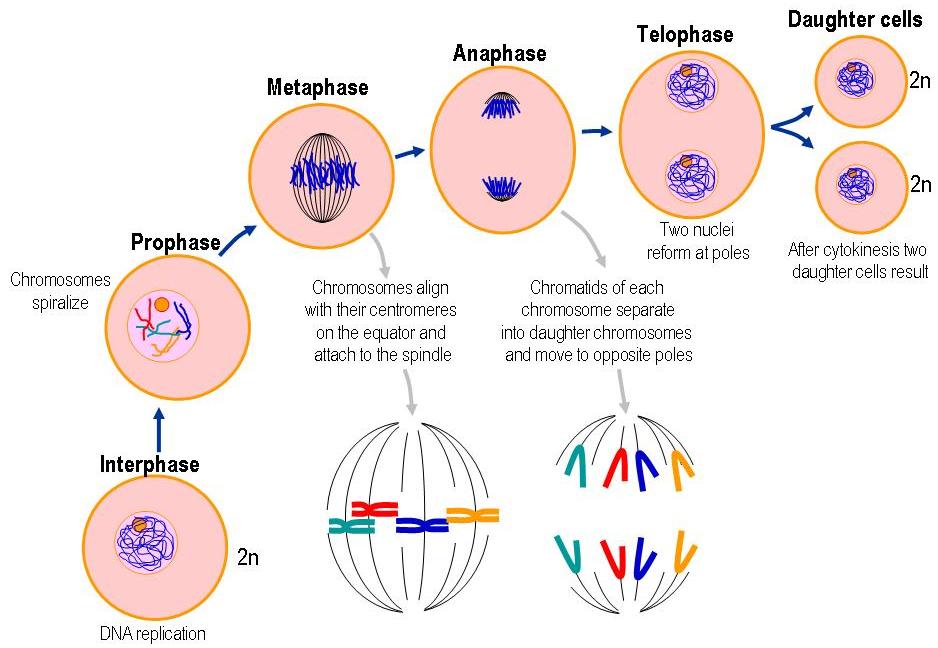
Homologous chromosomes are separated during meiosis I (MI), and sister chromatids are segregated during meiosis II (MII). During meiosis, a single round of DNA replication is followed by two consecutive chromosome segregations, meiosis I and II, which reduce the number of chromosomes by half. Meiosis gives rise to haploid gametes from diploid germ cells. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: The authors have declared that no competing interests exist. KC and GFV were supported by Institute for Protein Research, Osaka University. FK was supported by grant SFB34 by the Austrian Science Foundation. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: All relevant data are wishing the paper and its Supporting Information files.įunding: This work was supported by Japanese Society for Promotion of Science ( ) KAKENHI Grant Number 22125001, 22125002, 15H05973 and 16H04742. Received: JAccepted: NovemPublished: January 3, 2019Ĭopyright: © 2019 Challa et al. 
PLoS Genet 15(1):Įditor: Michael Lichten, National Cancer Institute, UNITED STATES Our results suggest that late meiotic prophase-I actively controls cohesin dynamics on meiotic chromosomes for chromosome segregation.Ĭitation: Challa K, Fajish V G, Shinohara M, Klein F, Gasser SM, Shinohara A (2019) Meiosis-specific prophase-like pathway controls cleavage-independent release of cohesin by Wapl phosphorylation. In the work presented here we confirmed that the prophase pathway is not suppressed during meiosis, at least in budding yeast and showed that this cleavage-independent release is regulated by meiosis-specific phosphorylation of two cohesin subunits, Rec8 and Rad61(Wapl) by two cell-cycle regulators, PLK and DDK. However, a previous study on budding yeast meiosis suggests the presence of cleavage-independent pathway of cohesin release during late prophase-I. In this context, it was long assumed but not properly investigated, that the prophase pathway for cohesin release would be specific to mitosis and would be safely suppressed during meiosis so as not to endanger essential connections between chromosomes. 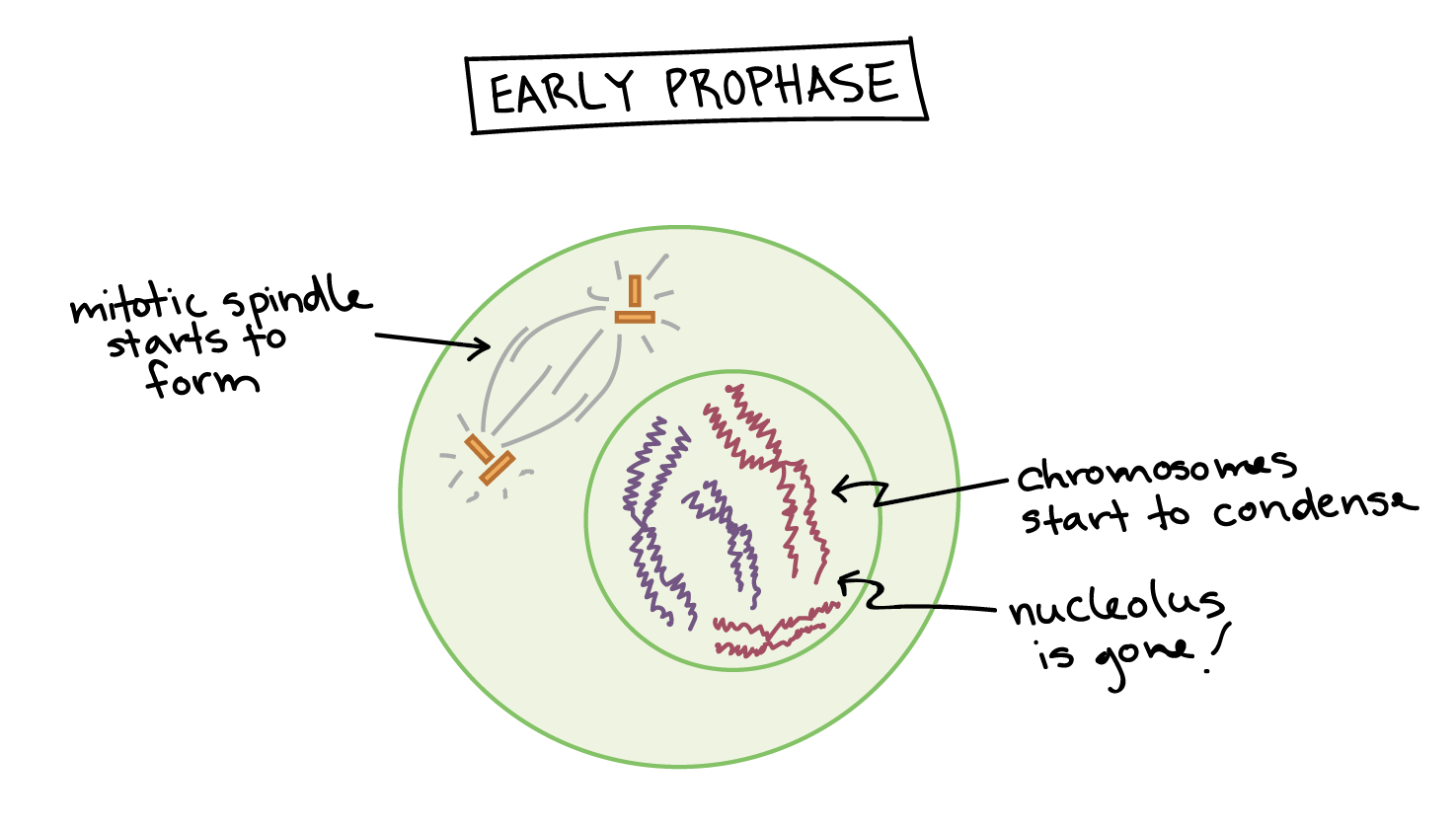
Indeed, it was shown that cohesive cohesin does not show an appreciable turnover during long periods in oocyte development. Therefore, it is currently believed that the physical connections between homologous chromosomes are maintained by meiotic cohesin with exceptional stability. Any failure in chromosome segregation has a detrimental impact. In meiosis the life and health of future generations is decided upon. We suggest that yeast uses this highly regulated cleavage-independent pathway to remove cohesin during late prophase-I to facilitate morphogenesis of condensed metaphase-I chromosomes. In yeast, meiotic cohesin release coincides with PLK-dependent compaction of chromosomes in late meiotic prophase-I. This process is similar to the vertebrate “prophase” pathway for cohesin release during G2 phase and pro-metaphase. Meiosis-specific phosphorylation of Rad61/Wpl1 and Rec8 by PLK and DDK collaboratively promote this release. 
This release requires, in addition to PLK, the cohesin regulator, Wapl (Rad61/Wpl1 in yeast), and Dbf4-dependent Cdc7 kinase (DDK). In this study, we confirmed that this release indeed accompanies the dissociation of acetylated Smc3 as well as Rec8 from meiotic chromosomes during late prophase-I. This decrease is seen in the absence of Rec8 cleavage and depends on condensin-mediated recruitment of Polo-like kinase (PLK/Cdc5). Paradoxically, reduced immunostaining signals of meiosis-specific cohesin, including the kleisin Rec8, were observed on chromosomes during late prophase-I of budding yeast. It was assumed that, unlike the situation in mitosis, chromosome arms retain cohesion prior to onset of anaphase-I. Sister chromatid cohesion on chromosome arms is essential for the segregation of homologous chromosomes during meiosis I while it is dispensable for sister chromatid separation during mitosis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
